Report on the ECA “Equipment Qualification Forum”
This year's ECA "Equipment Qualification Forum" focused on the use of the ECA Good Practice Guide Qualification and Validation - A guide to effective qualification based on Customer - Supplier Partnership, Version 3.0.
At the forum, authors and users of the guide explained its content and possible applications to 25 participants, as well as the direction in which the guide could develop. Below is an overview of selected contributions from the two event days.
At the start of the event, the current head of the ECA Validation Group, Ralf Gengenbach, gave an overview of the Validation Group and its sub-groups, as well as the guide itself. The group's objectives are:
- To offer practical solutions in the field of qualification and validation
- To present case studies
- To provide examples
Before discussing the guide itself, Ralf Gengenbach posed the rhetorical question, "Why another guide?" He then answered his own question:
- The "how-to" of device qualification is still a challenge when it comes to details
- The integration of engineering (Good Engineering Practice; GEP) into equipment qualification (GMP) is often still missing or has not yet been implemented as comprehensively as might be possible
- Cooperation between suppliers and customers can still be improved
- How do we focus on the critical aspects in the context of risk assessment?
This is where the ECA Good Practice Guide comes in. It aims to outline practical solutions, incorporate case studies, and provide examples of best practices in the spirit of "lean qualification." The guide links the requirements of Annex 15 of the EU GMP Guidelines with the FDA Process Validation Guidance and the approach of ASTM Guideline 2500. But how does the ECA Good Practice Guide differ from ISPE's "Commissioning and Qualification Guide"? The ISPE Guide focuses on the process flow, while the ECA Guide focuses on the different parties and their tasks in equipment qualification.
Finally, Ralf Gengenbach gave an outlook on the planned further development of the guide.
Recommendation
29/30 September 2026
Process Validation - Live Online Training
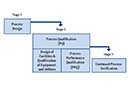
Ralf Gengenbach, who, in addition to his leadership role in the ECA Validation Group, is also the managing director of gempex, revisited the topic of good engineering practice in a separate presentation after emphasizing its importance in his introduction. Right at the beginning of his presentation, he made it clear that 80% of qualification activities can be mapped with GEP. Without GEP, it is not possible to demonstrate that a system is suitable, which is the goal of qualification. Ralf Gengenbach sees the major advantages of GEP in the clearly defined milestones and the clearly defined workflow.
Both lead to clear project plans. Using practical examples, he then showed possible implementations of the most important GEP documents:
- Process documents
- Engineering documents
- Management requirements
- Commissioning documents
And what was still causing problems? This is shown in Figure 1.

Figure 1: Typical Pitfalls - Critical Topics (Summary)
The first day ended with a presentation by Dr Timo Kretzschmar from TikreSolution. His topic: "AI - "Prompt": Artificial Intelligence, qualify yourself - or how else". Instead of an introduction, he briefly mentioned that there is currently no one who has a complete overview of the field of artificial intelligence. Artificial intelligence is currently developing at a highly dynamic pace. His presentation was intended to stimulate discussion. He then used a graphic to show the connection between artificial intelligence, machine learning, and "deep learning".
Starting with the EU's AI Act, he then moved on to regulated environments, in particular GMP. He supported all his statements with further links. He named the following classes of artificial intelligence that occur in a GMP environment:
- Static deterministic models
- Dynamic adaptive models
- Generative and probability models
In his opinion, a dynamic adaptive model is viewed critically by regulatory authorities, and he considers the use of generative and probability models unacceptable in critical GMP applications. Timo Kretschmar then gave an overview of the Annex 22 draft, which also included many links for further reading on the topic. He sees the possibility of using artificial intelligence to support the validation of computerised systems in the future. Instead of a case study, he concluded by providing information on an artificial intelligence-supported document management system.
Day two began with surveys on the ECA Good Practice Guide itself. Ralf Gengenbach provocatively asked whether such a guide is still valuable in the age of artificial intelligence. All participants still considered the guide necessary, with some expressing a desire for further improvements. In particular, more case studies and more details on individual equipment were requested, in addition to forms and templates.
Maik Guttzeit then spoke about user requirements. His equally provocative presentation title was "User Requirement Specification - the Biggest Risk - or the Greatest Opportunity."
He began with a slide on which he listed the contents of a user requirement:
- All technical details of the entire machine or system
- It is better to include more documents related to a user request (e.g., internal SOPs) than to forget something
- The project should be adapted as closely as possible to customer standards (such as welding specifications)
- Since suppliers often do not understand the expectations of pharmaceutical companies, user requirements serve as a guideline for suppliers
Then Ralf Eigenpath, head of the ECA Validation Group, intervened and said that a user requirement should not actually be exactly that. And then it became clear that Maik Guttzeit had used the first slide as a rhetorical device to show that this is often still the case.
In his opinion, user requirements should:
- Focus on the absolutely essential requirements necessary to ensure the suitability of equipment
- Be short and concise, as too much complexity and unnecessary details carry the risk that the user requirements will not be read at all ("too long - didn't read")
- Involve suppliers (they are the experts for their products)
- Be the leading document for the pharmaceutical company and form the basis for the use of equipment or systems for GMP-compliant production
He then divided what a user needs into
- GMP-relevant, which goes into the user requirements
- Everything else, which goes into the technical specifications
- Budget issues and delivery deadlines go into other documents ("Terms/Conditions")
According to Guttzeit, user requirements should not contain any technical solutions (e.g., diaphragm valves as a requirement for product-contacting lines).
But how do you determine GMP requirements? Maik Guttzeit also provided some tips here:
- Does the requirement affect product quality without being noticed and could it harm patients?
- Is the requirement necessary throughout the entire life cycle of the equipment/system and can it be verified?
- Does the device/system fail to meet the suitability for use requirement if the requirement is not met?
- Is the requirement the only possible solution (no alternative or workaround possible)?
- Has this requirement already been successfully used in other GMP applications?
- Are there direct links to binding guidelines (GMP guidelines, Code of Federal Regulation, ICH quality guidelines)?
If all answers are yes, you have defined the GMP requirement, according to Maik Guttzeit.
Recommendation
14-16 October 2026
Advanced Level: Trending of Process Data for OPV/CPV - Live Online Training
He then discussed possible implementations in an "integrated qualification concept" (see Figure 2).

Figure 2: Integrated Qualification Concept
Finally, he addressed the use of artificial intelligence in creating user requirements. He sees the tool as an aid, but warns against using it too uncritically (are all aspects covered, be careful with confidential company information in open models). His conclusion on the use of artificial intelligence in creating user requirements: Use your brain!
The last presentation was on the topic of "fleet management." It was given by two speakers from Hoffmann-La Roche, Dr Christian Siegmund and Sebastian Faller. The idea behind "fleet management" is to achieve modular standardization across the "fleet":
- Modular strategy on a small scale, then scaling out
- Modular standardisation through off-the-shelf equipment
- A modified batch and sampling strategy to become more efficient and reduce process losses
- Improvement of product quality through modular standardised equipment, resulting in more stable processes due to directly comparable process data and fewer human interventions and errors
So what exactly is a "fleet"? It is a group of equivalent systems in terms of design and installation, based on the same specifications across multiple manufacturing facilities and locations. Controlled by a central pharmaceutical quality system throughout the entire life cycle of the equipment. Equivalence means that manufacturing systems remain identical in terms of cGMP use in critical aspects (specifications, design, functions, operation, qualification). The consideration of equivalence encompasses many aspects:
- Supplier relationship/user requirements (suppliers must understand the concept)
- Equivalence of components (filters, valves, etc.)
- Automation/software (software versions, tests, etc.)
- Qualification and validation (protocols, test standards, templates, etc.)
- SOPs (training standards, etc.)
- Product contact (primary packaging materials, closures)
- Equipment preparation (cleaning procedures, clearing the line, etc.)
- Process (fermentation, filling, etc.)
The vision is to design one piece of equipment and then build many more. This equivalence also makes it easier to scale up and transfer to other locations, as the equipment is always the same. Implementing a global "fleet" requires appropriate management with a strategic level at the top, an operational level in the middle, and local project and site teams for implementation (see Figure 3).

Figure 3: Global Fleet Management
Of course, a tightly structured change management system must also be in place to maintain equivalence or to remove individual equipment from the fleet if equivalence can no longer be guaran teed. As already mentioned, all of this takes place within a pharmaceutical quality management system, into which fleet management must be integrated. Another important element within the "fleet" is knowledge management, which is necessary in order to be able to permanently map the above-mentioned points.
And what are the real benefits of such a "fleet"?
Siegmund and Faller saw savings of 15% in the finished product area for design, installation, engineering, and maintenance costs for a second and third machine within a fleet. They also saw a saving of four months at the supply end due to a shorter concept phase and savings of around two weeks for FAT tests on the second and third machines due to parallel processing.
They see even greater savings potential in the area of active ingredient production. They assume a cost reduction of 60% at location B in terms of automation compared to location A. And for picking and qualification activities, they expect savings of 35% between site A and site B, even with more equipment delivered to site B. In their summary, the speakers talk about a possible "game changer" if the following are in place:
- Very good knowledge of the equipment and processes
- Committed support from senior management
- High discipline among all those involved
- Good communication o Support and acceptance from the authorities
Conclusion: The ECA Equipment Qualification Forum provided a platform for exchange, particularly between participants and speakers. The topic of equipment qualification remains highly relevant. Version 3.0 of the ECA Good Practice Guide "Qualification and Validation - A guide to effective qualification based on Customer - Supplier Partnership" has proven its worth. In its further development, more case studies, more details on individual equipment, and more sample documents were requested. And that is exactly what the working group will be working on in 2026.
| The guide is available free of charge to members of the |
About the Author
Sven Pommeranz is Operations Director and organises and conducts courses and conferences on behalf of the ECA Academy in the area validation.



